These ratios by themselves may not seem particularly interesting or informative however, if we take a ratio of these ratios, we obtain a useful and possibly surprising result: a small, whole-number ratio. 
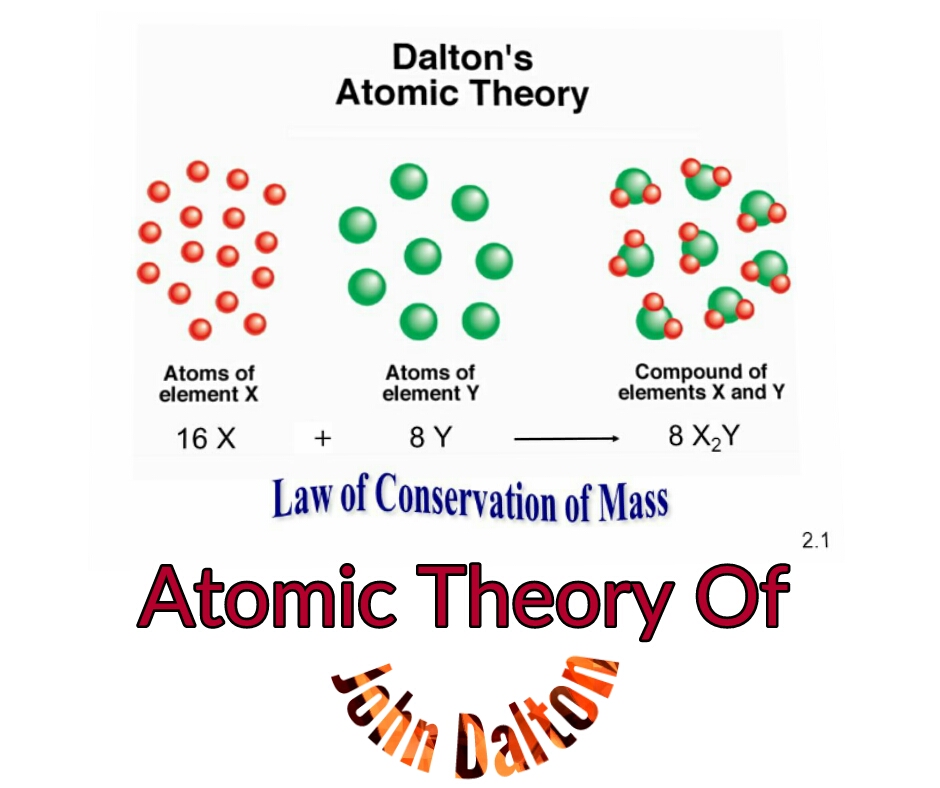
This can be gleaned from the third postulate in Daltons series. For example, copper and chlorine can form a green, crystalline solid with a mass ratio of 0.558 g chlorine to 1 g copper, as well as a brown crystalline solid with a mass ratio of 1.116 g chlorine to 1 g copper. Study with Quizlet and memorize flashcards containing terms like Which of the following reactions is possible according to Daltons atomic theory, The law of conservation of mass states that mass is neither created nor destroyed during a chemical reaction. 
The postulate of Dalton's atomic theory, 'Atoms of different elements combine in fixed ratios to form compounds,' results from the law of definite proportions. The law of multiple proportions states that when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small, whole numbers. The law of definite proportions states that the given chemical compound always contains its component elements in a fixed ratio. For example, there are many compounds other than isooctane that also have a carbon-to-hydrogen mass ratio of 5.33:1.00.ĭalton also used data from Proust, as well as results from his own experiments, to formulate another interesting law. That is, samples that have the same mass ratio are not necessarily the same substance. It is worth noting that although all samples of a particular compound have the same mass ratio, the converse is not true in general.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
